 |
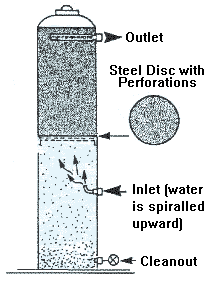
The most common, oldest and relatively inexpensive method used to
disinfect water is chlorination. A chemical feed pump continuously
dispenses chlorine into the water supply. Chlorine, an
oxidizing agent, kills most bacteria and some viruses. In the proper
concentrations and under adequate exposure time, chlorine is an
excellent disinfectant.
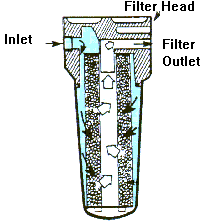
Chlorine reacts with certain metals and organic matter in the water
to form hazardous, chlorinated, organic chemicals. Use of an activated
carbon filter after chlorination will remove excess chlorine and
limited amounts of any chlorine compounds formed.
Chlorination may also oxidize
and remove some color and odor causing substances, including
iron and hydrogen sulfide.
|